Foods | Free Full-Text | Towards Substitution of Hexane as Extraction Solvent of Food Products and Ingredients with No Regrets

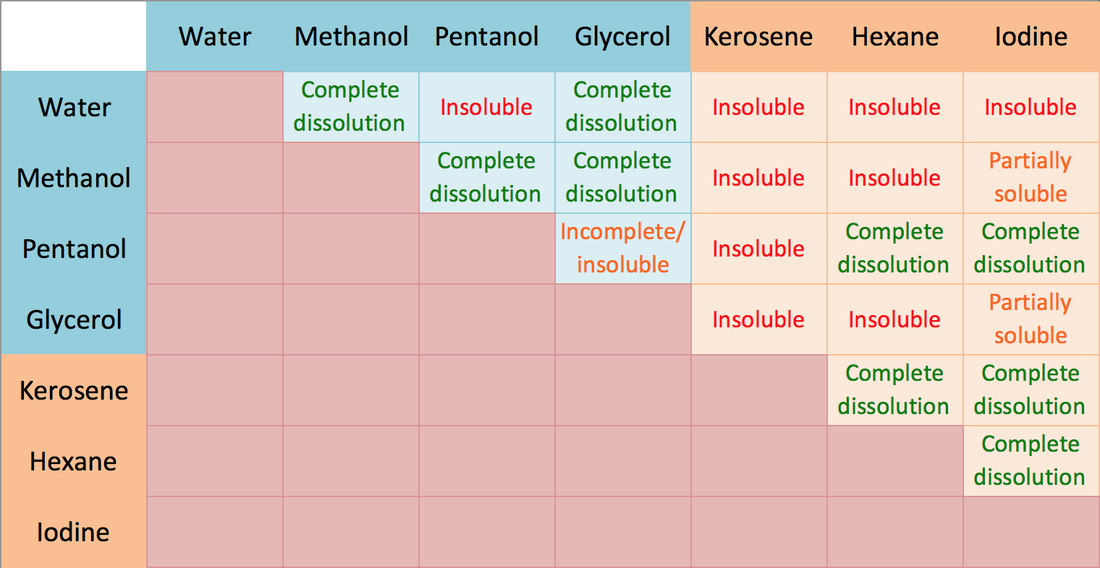
SOLVED:Water is a polar solvent and hexane (C6 H14) is a nonpolar solvent. In which solvent is each of the following, which is found or used in the body, more likely to

1) Rank trans-stilbene, benzoic acid, and fluorenone by polarity (least polar to most polar). Provide an explanation for your proposed ranking. (2) Hexane, ethyl acetate and acetone will be used as

Question Video: Selecting the Statement Which Explains Why Hexane and Water Are Not Miscible | Nagwa