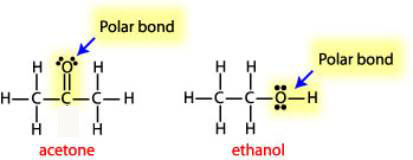
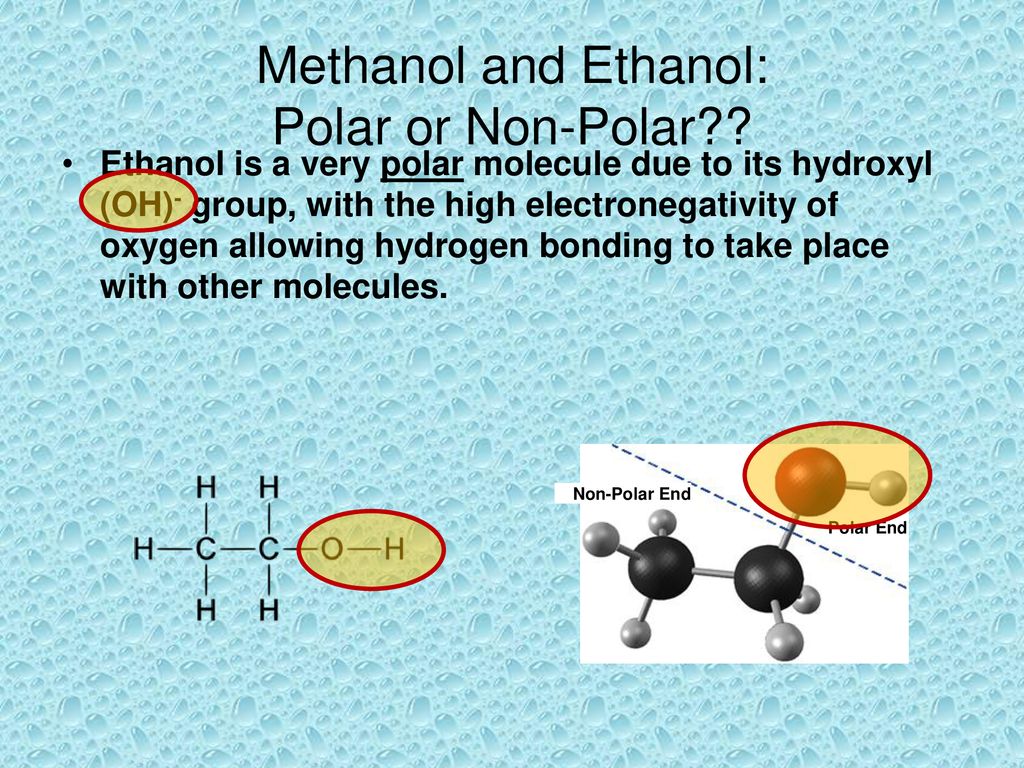
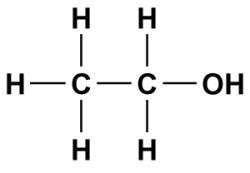
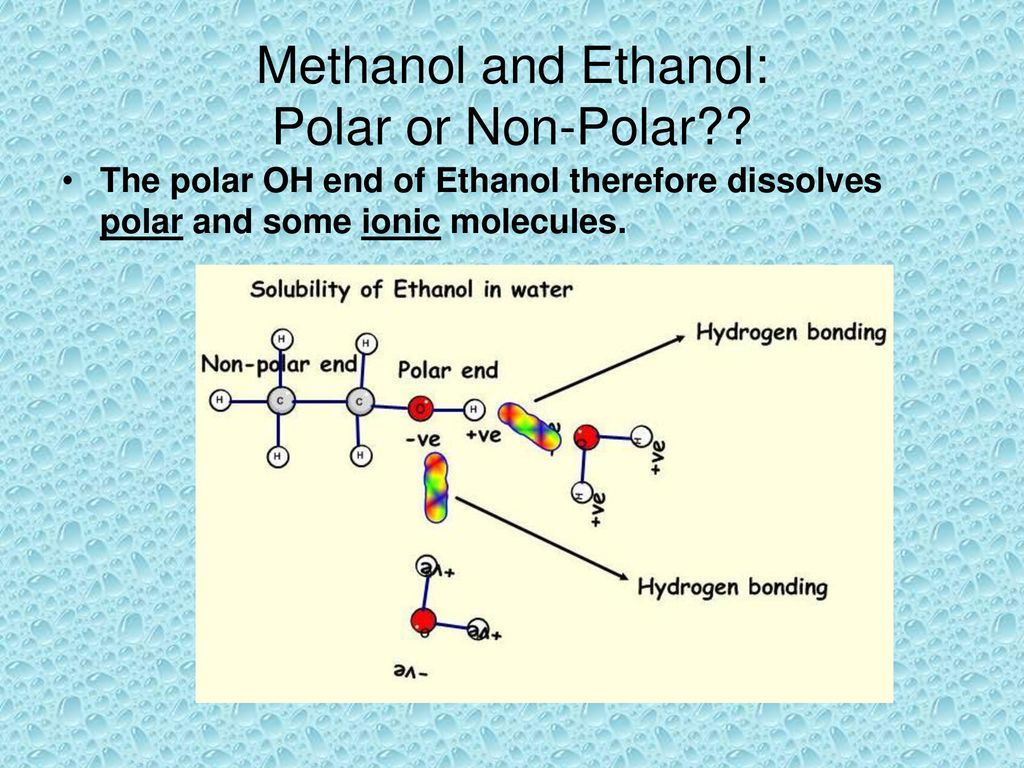
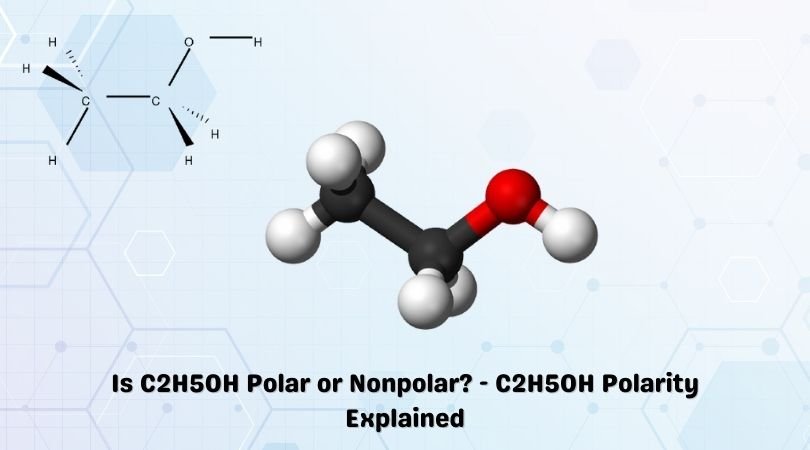
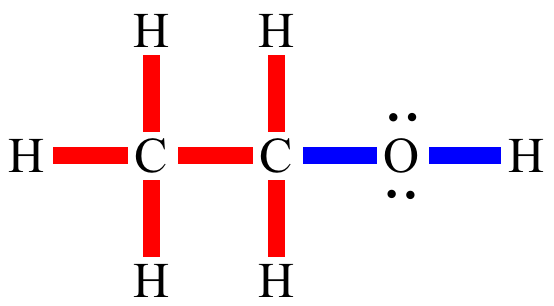
a) Are there polar bonds in Ethanol? (Show EN for each of the bonds in the molecule) b) What is the geometric shape of Ethanol (VSEPR)? c) Is this molecule polar or

SOLVED: Which of the following statements best accounts for the difference in boiling point between these two molecules? H C-H H= OH Ethane Ethanol -89 %C +78 'C Boiling Point Select one: