
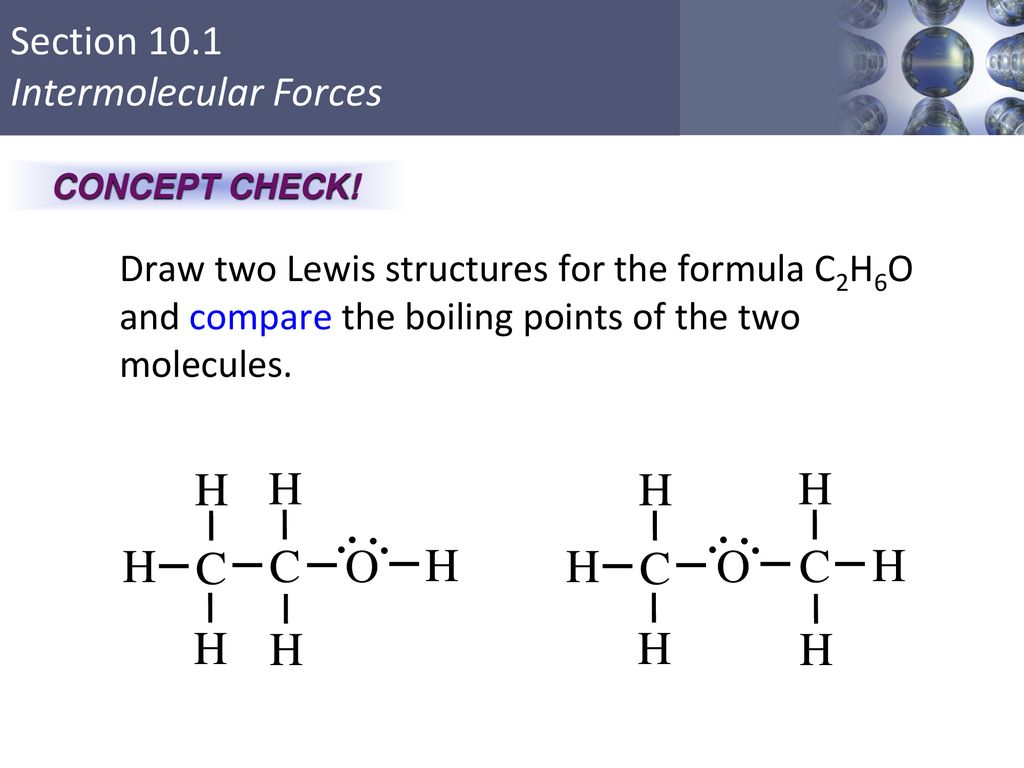
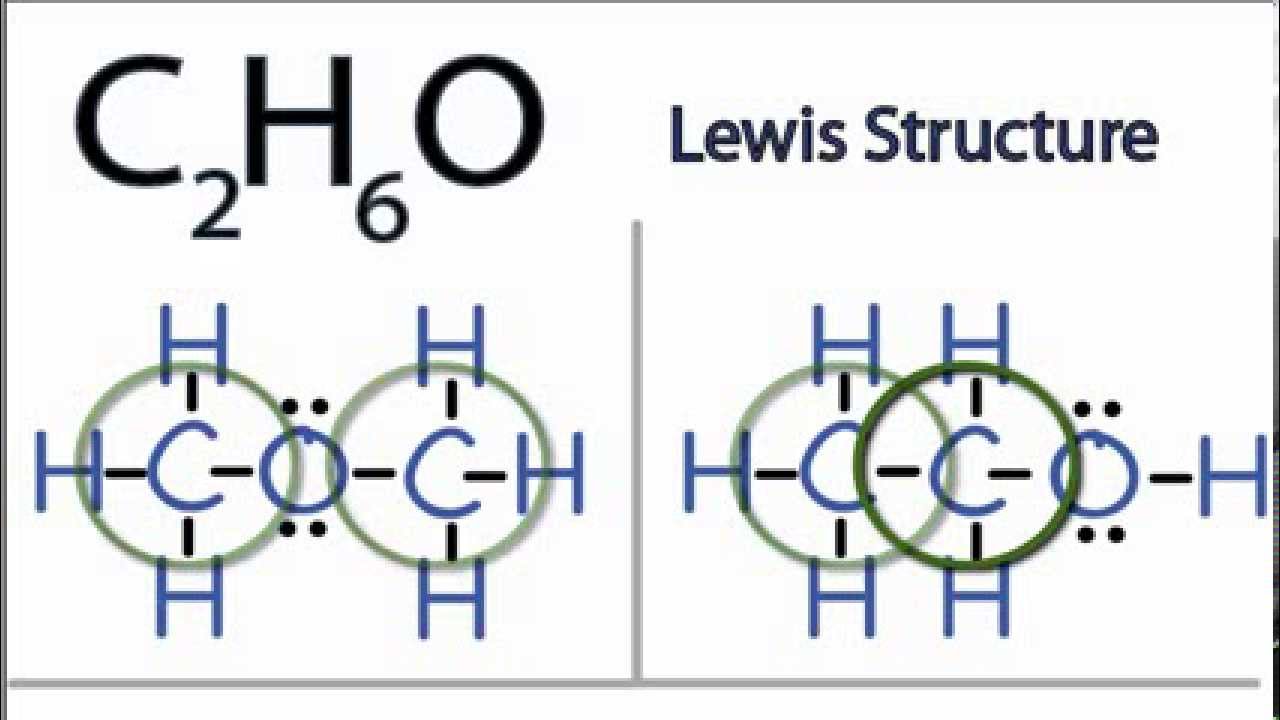
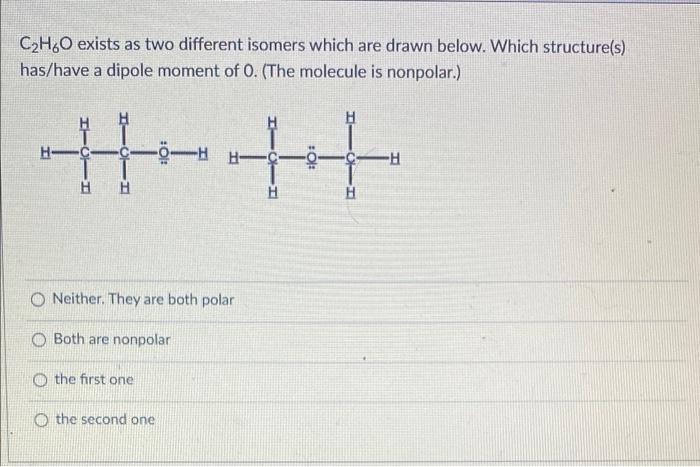
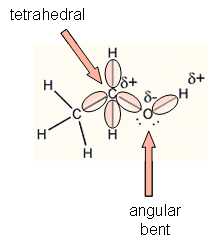
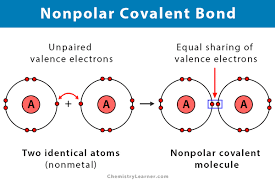
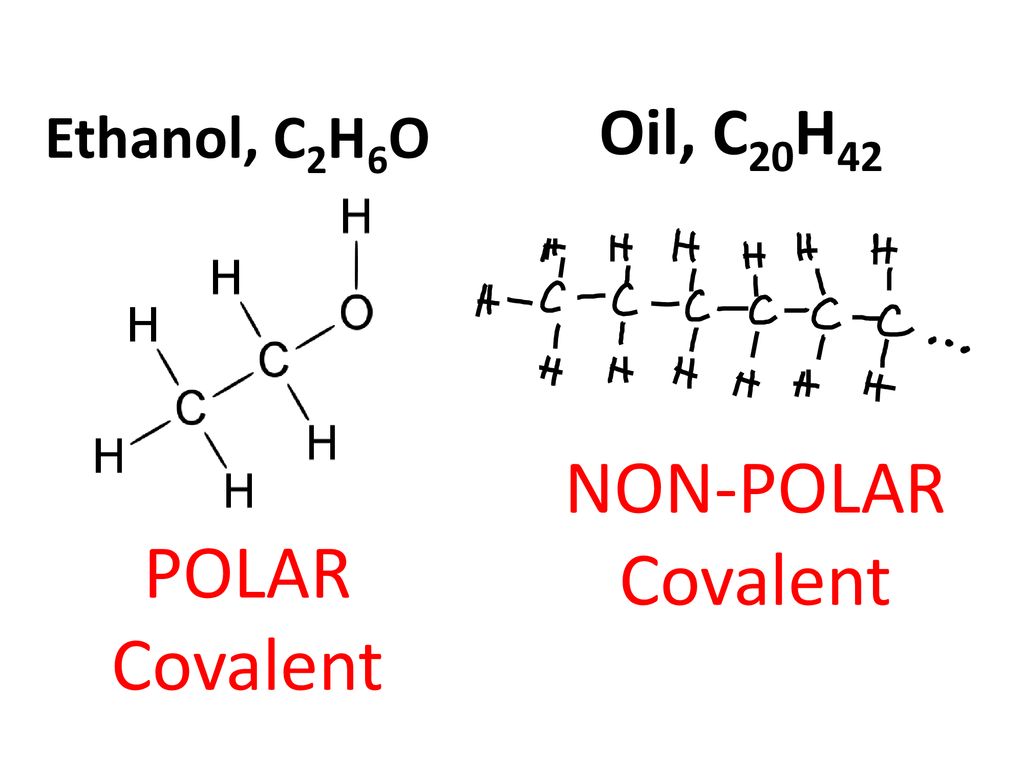
Alcohols are organic molecules that have a hydroxyl (-OH) group that is connected by a single bond to a carbon atom. Let's consider the molecule ethyl alcohol (C2H6O). a. What is the

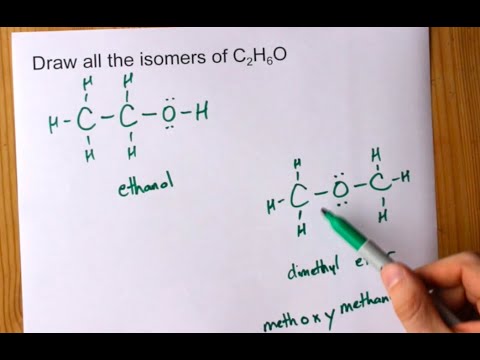
SOLVED: a. There are two forms of C2H6O, one of which is a liquid at room temperature and the other is a gas. (More than one structure for the same molecular formula















![Biology] Polar Bonds - General Discussion - Neowin Biology] Polar Bonds - General Discussion - Neowin](https://cdn.neowin.com/forum/uploads/post-107847-1188869267_thumb.jpg)